Pharmaceutical intermediates are chemical substances produced during the multi-step synthesis of active pharmaceutical ingredients (APIs). While they themselves do not have therapeutic effects, they are crucial steps in the synthesis of APIs. Intermediates undergo various chemical transformations before finally being converted into APIs. These compounds ensure that the final drug product meets the necessary efficacy, safety, and quality standards.
V&S Chem is a professional manufacturer and supplier in China, specializing in providing high-quality pharmaceutical intermediates. We meticulously manufacture these intermediates to meet the stringent standards required for API production. We understand that any impurities in intermediates can jeopardize the entire drug manufacturing process; therefore, we strive to ensure that all intermediates have the highest purity and consistency.
Intermediates act as the molecular “stepping stones” in the API synthesis process. Their function in drug development is multi-faceted, and their quality plays a direct role in the overall efficiency, safety, and scalability of pharmaceutical production.
Enabling Complex Synthesis: Intermediates allow for multi-step chemical reactions, ensuring that the final API is synthesized with the precision required for therapeutic efficacy.
Maintaining Consistency: The purity and consistency of intermediates are essential for producing APIs that meet strict regulatory standards, ensuring the quality of the final product.
Enhancing Production Efficiency: By breaking the synthesis process into manageable steps, intermediates allow for early detection of issues, reducing waste and costs, while improving overall efficiency in production.
Supporting Scalability: Intermediates are essential for large-scale production of APIs, ensuring consistent and reliable quality that can meet the high demands of the pharmaceutical industry.
Regulatory Compliance: At V&S Chem, we understand the importance of adhering to regulatory standards. Our intermediates are produced in strict compliance with Good Manufacturing Practices (GMP) to facilitate the successful approval of the final API by regulatory authorities.

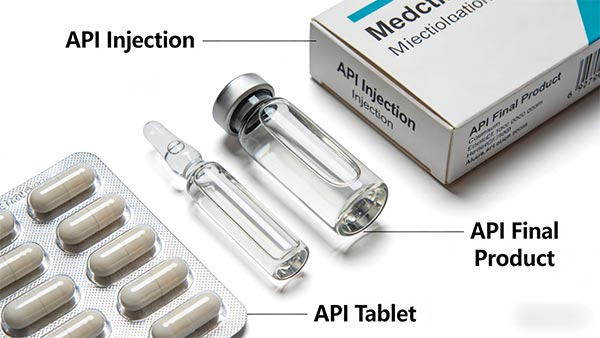


While both intermediates and APIs are integral to drug production, they play distinct roles. Understanding the key differences between these two components is crucial in appreciating the pharmaceutical manufacturing process:
| Aspect | Active Pharmaceutical Ingredients (APIs) | Pharmaceutical Intermediates |
| Function | APIs are the final, therapeutic agents that produce a health effect. | Intermediates are transitional compounds that eventually lead to the formation of APIs. |
| Production Stage | APIs are produced at the final stage of synthesis and are ready for drug formulation. | Intermediates are produced at earlier stages, and they serve as precursors in the synthesis process. |
| Purity Requirements | APIs must meet stringent purity standards to ensure their therapeutic effect. | Intermediates are also pure but may have slightly relaxed standards as they will undergo further processing. |
| Regulatory Oversight | APIs are heavily regulated by authorities like the FDA and EMA due to their direct impact on human health. | Intermediates are subject to less regulation, but they still require careful quality control to prevent contamination in the final API. |
| Storage and Stability | APIs require stable storage conditions to maintain their effectiveness over time. | Intermediates often have a shorter shelf life and are typically stored under specific conditions for immediate use in further production. |
The quality of pharmaceutical intermediates directly affects the quality of the final API. Even minor impurities in an intermediate can lead to inefficacy or safety concerns in the final drug product. Therefore, controlling the synthesis, handling, and storage of intermediates is crucial to ensuring that APIs are safe and effective for patient use.
Ensures Consistency: Consistent quality of intermediates leads to consistent API production, guaranteeing that each batch of medication performs as expected.
Reduces Risks: Controlling impurities at the intermediate stage helps prevent contamination in the final product, reducing the risk of side effects or adverse reactions.
Facilitates Regulatory Approval: Proper handling and production of intermediates are critical to obtaining regulatory approval for the final API. This helps in meeting international standards and gaining access to global markets.
At V&S Chem, we are committed to providing the highest-quality pharmaceutical intermediates, ensuring that our customers in the pharmaceutical industry can produce safe, effective, and consistent APIs. Here’s why pharmaceutical manufacturers trust us:
High-Quality Standards: We adhere to rigorous quality control processes to ensure that our intermediates meet the highest purity standards.
Expertise and Experience: With years of experience in the chemical industry, we specialize in supplying high-quality intermediates for pharmaceutical applications.
Regulatory Compliance: All of our intermediates are produced in compliance with GMP standards, ensuring that they meet international regulatory requirements.
Customized Solutions: We understand that each pharmaceutical company has unique needs. That’s why we offer tailored solutions to meet the specific requirements of API production.
Pharmaceutical intermediates are indispensable in the journey of transforming raw materials into effective drugs. They play a critical role in enabling the synthesis of APIs, maintaining drug quality, and ensuring regulatory compliance. At V&S Chem, we are dedicated to supporting pharmaceutical companies with the highest-quality intermediates, produced to meet the strictest standards for safety, efficacy, and consistency. By choosing us as your supplier, you can be confident that you are sourcing top-quality intermediates that will contribute to the successful production of your APIs and, ultimately, the better health of patients worldwide.
If you're looking for reliable, high-quality pharmaceutical intermediates, don’t hesitate to reach out to us today.

